
Of care known as the EXTREME regimen,” said Dr. “It is exciting to see the full results from this trial, which is theįirst study to show superior overall survival over the current standard ≥20 and CPS ≥1 compared with the EXTREME regimen, the current standard Patients whose tumors expressed PD-L1 with Combined Positive Score (CPS) Results of an interim analysis were presented at theĮuropean Society for Medical Oncology (ESMO) 2018 Congress andĭemonstrated superior OS outcomes for KEYTRUDA in combination withĬhemotherapy in the total population and KEYTRUDA monotherapy in PD-L1 expression and the KEYTRUDA monotherapy study arm in the total The first-time presentation of certain overall survival (OS) hypothesesįrom the KEYTRUDA in combination with chemotherapy study arm based on Neck squamous cell carcinoma (HNSCC) at the 2019 American Society ofĬlinical Oncology (ASCO) Annual Meeting (Abstract #6000). Therapy, as monotherapy and in combination with chemotherapy, for theįirst-line treatment of patients with recurrent or metastatic head and 
Phase 3 KEYNOTE-048 trial investigating KEYTRUDA, Merck’s anti-PD-1 Today announced the presentation of the final analysis of the pivotal KENILWORTH, N.J.–( BUSINESS WIRE)–Merck (NYSE:MRK), known as MSD outside the United States and Canada, Pembrolizumab alone group, 32 (12%) in the pembrolizumab with chemotherapy group,Īnd 28 (10%) in the cetuximab with chemotherapy group.Survival Benefit Now Observed with KEYTRUDA in Combination with Chemotherapy in All Patient Populations (Regardless of PD-L1 Expression) and with KEYTRUDA Monotherapy in Patients Whose Tumors Expressed PD-L1 at CPS ≥1ĭata Presented Today at 2019 ASCO Annual Meeting Adverse events led to death in 25 (8%) participants in the Of 276 in the pembrolizumab with chemotherapy group, and 239 (83%) of 287 in the cetuximab In 164 (55%) of 300 treated participants in the pembrolizumab alone group, 235 (85%) At final analysis, grade 3 or worse all-cause adverse events occurred Nor pembrolizumab with chemotherapy improved progression-free survival at the second Versus cetuximab with chemotherapy in the total population (13♰ months vs 10♷ months, HR 0♷7, p=0♰034) at the second interim analysisĪnd in the CPS of 20 or more population (14♷ vs 11♰, 0♶0, p=0♰004) and CPS of 1 or more population (13♶ vs 10♴, 0♶5, p<0♰001) at final analysis. Pembrolizumab with chemotherapy improved overall survival More population (12♳ vs 10♳, 0♷8, p=0♰086) and was non-inferior in the total population (11♶ (median 14♹ months vs 10♷ months, hazard ratio 0♶1, p=0♰007) and CPS of 1 or Overall survival versus cetuximab with chemotherapy in the CPS of 20 or more population At the second interim analysis, pembrolizumab alone improved With chemotherapy (n=300) of these, 754 (85%) had CPS of 1 or more and 381 (43%) Pembrolizumab alone (n=301), pembrolizumab with chemotherapy (n=281), or cetuximab Findingsīetween April 20, 2015, and Jan 17, 2017, 882 participants were allocated to receive This study is registered at, number NCT02358031. Safety was assessed in the as-treated population (all participants who received at Were obtained when statistical testing was completed for that hypothesis this occurredĪt the second interim analysis for 11 hypotheses and at final analysis for three hypotheses. The definitive findings for each hypothesis Of pembrolizumab alone and pembrolizumab with chemotherapy versus cetuximab with chemotherapyįor overall survival in the total population. Of 1 or more, and total populations and non-inferiority (non-inferiority margin: 1♲) Overall survival and progression-free survival in the PD-L1 CPS of 20 or more, CPS 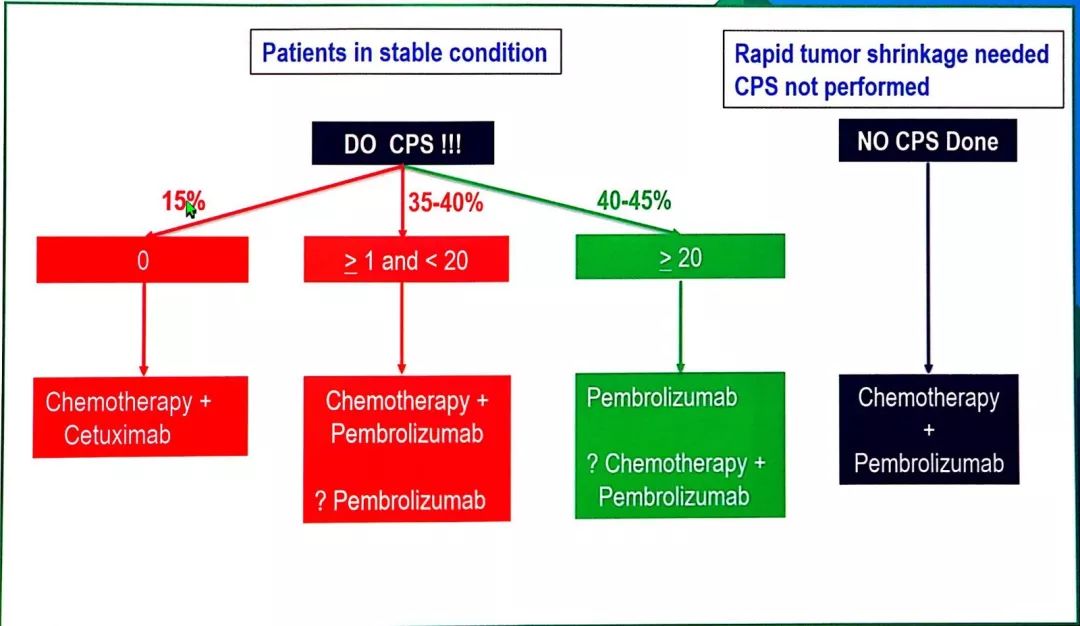
There were 14 primary hypotheses: superiority of pembrolizumabĪlone and of pembrolizumab with chemotherapy versus cetuximab with chemotherapy for Radiographically confirmed disease progression or death from any cause, whicheverĬame first) in the intention-to-treat population (all participants randomly allocated To death from any cause) and progression-free survival (time from randomisation to The primary endpoints were overall survival (time from randomisation To the PD-L1 combined positive score (CPS) results PD-L1 positivity was not requiredįor study entry. Investigators, participants, and representatives of the sponsor were masked Investigators and participants were aware of treatmentĪssignment. (pembrolizumab with chemotherapy), or cetuximab plus a platinum and 5-fluorouracil Were stratified by PD-L1 expression, p16 status, and performance status and randomlyĪllocated (1:1:1) to pembrolizumab alone, pembrolizumab plus a platinum and 5-fluorouracil Incurable recurrent or metastatic HNSCC done at 200 sites in 37 countries. KEYNOTE-048 was a randomised, phase 3 study of participants with untreated locally The Lancet Regional Health – Western Pacific.The Lancet Regional Health – Southeast Asia.The Lancet Gastroenterology & Hepatology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
